Numerous researchers have identified the potential impact of clinically useful blood biomarkers associated with TBI on ED patient care
Numerous researchers have identified the potential impact of clinically useful blood biomarkers associated with TBI on ED patient care
In a comprehensive review paper in the publication Proteomics - Human Diseases and Protein Functions, Exploring the Role of Biomarkers for the Diagnosis and Management of Traumatic Brain Injury Patients, Dr. Papa discusses various potential gains of biomarkers for TBI. These gains could include improving patient stratification by injury severity; detecting microinjuries to the brain that cannot be seen by CT; advancing assessment of intoxicated, unconscious, sedated, or polytrauma patients; and identifying patients at risk of developing long-term sequelae.1
In a 2019 article in The Lancet Neurology, Yue et al proposes that the use of biomarkers could improve timing and accuracy of diagnosis, as well as guiding treatment and surveillance strategies.2
The potential impact of using biomarkers in the care of possible TBI patients is further described by Bazarian et al. In a 2018 article in The Lancet Neurology, the investigators maintain that up to one-third of head CT scans done in the acute setting to assess TBI could be avoided, with a very low false-negative rate. Moreover, the clinical use of biomarkers could reduce unnecessary head CT scans with their attendant radiation exposure and cost.3
In a 2018 article in The Western Journal of Emergency Medicine, Michelson et al argues that reducing unnecessary CT scans in the ED could improve ED performance, accelerate patient disposition, and reduce length of stay.4
Korley et al underscore the importance of transforming TBI care through diagnostic advancement. In a 2015 article in The Journal of Head Trauma Rehabilitation, the authors assert: “…the need for novel diagnostics to optimize the accuracy, efficiency, and cost-effectiveness of [TBI] evaluations constitutes an important public health concern.”5
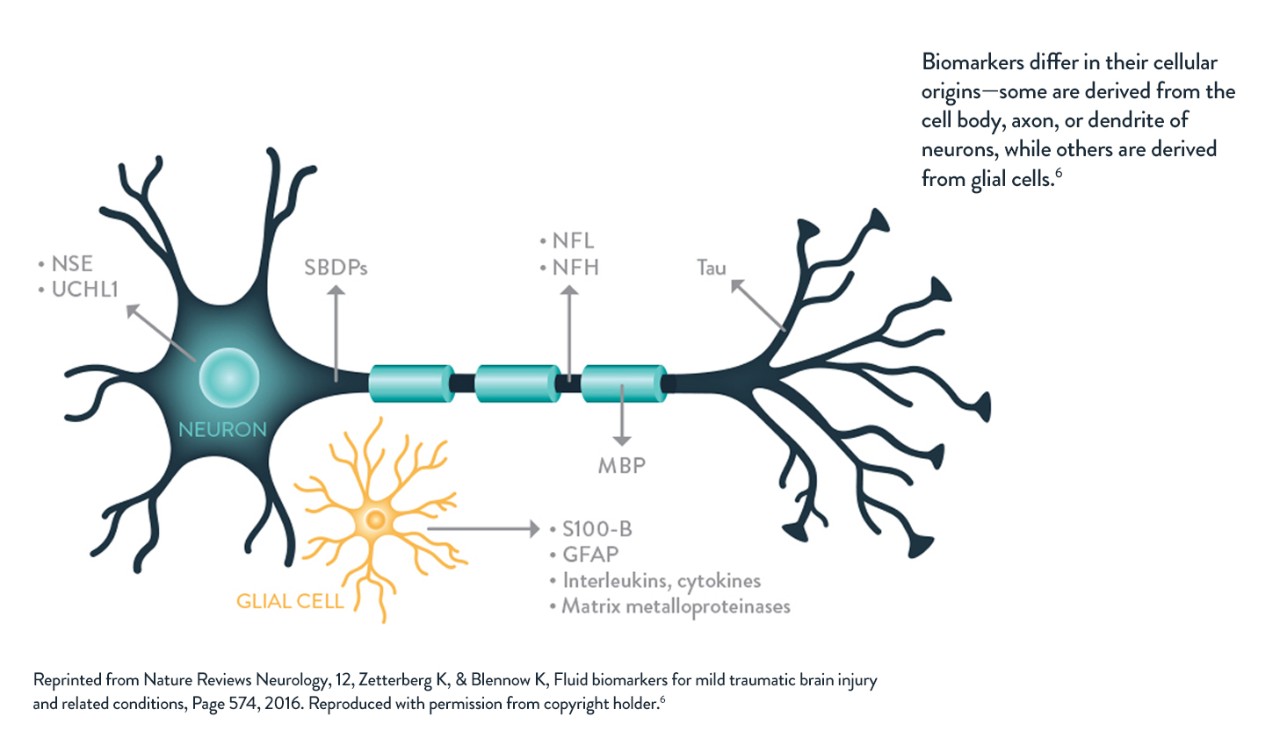
TBIs affect different pathophysiologic pathways in the central nervous system. Brain biomarker assays may detect brain specific proteins and provide objective, quantitative information to aid the TBI assessment.
“Following a head injury, cerebrovascular endothelial cells often incur mechanical deformation, as well as shear stress injury, leading to increased tight junction permeability and leakage of molecules across the blood-brain barrier. Theoretically, these events would lead to increased concentrations of CNS-derived molecules in the blood and of blood-derived molecules in the CSF.” 6
– Zetterberg, Blennow
For biomarkers to have diagnostic value in the ED, certain traits are considered essential.7,8



Biomarker levels must be detectable in peripheral blood shortly after injury onset



The sensitivity of the biomarker must be high enough to detect mild injury



Elevated biomarker levels must be specific to head trauma
Stay on top of biomarker research.


Ubiquitin C-terminal hydrolase-L1 (UCH-L1), a protein involved in the metabolism of ubiquitin, has been shown to be elevated in serum shortly after head injury. Increases in blood UCH-L1 have been detected in the serum of mild and moderate TBI patients within an hour of injury.9 Levels taken within 4 hours of injury were significantly higher in those with TBI lesions on CT than those with a normal intracranial appearance at CT.9 Blood levels of UCH-L1 have been shown to discriminate between mTBI patients and patients without head injuries.9 UCH-L1 levels were much higher in patients who required neurosurgical intervention.9
Classification performance for detecting intracranial lesions on CT at a UCH-L1 cutoff level of 0.09 ng/mL yielded a sensitivity of 100% (95%CI 88–100), a specificity of 21% (95%CI 13–32) and a negative predictive value of 100% (76–100).9



Neuron specific enolase (NSE) is an enzyme found in neuronal cell bodies throughout the nervous system that has been shown to be elevated in serumafter head injury.9 Several reports describe the utility of NSE as a marker of neuronal injury.9-11 A reported drawback of using NSE as a specific marker for TBI is its abundant expression in red blood cells,8 which has prompted researchers to use a hemolysis correction when measuring NSE in blood.12
One study examined the performance of serum NSE concentration within 3 hours after injury to identify intracranial lesions in mild TBI and found a sensitivity of 56% and a specificity of 77%.13



Glial fibrillary acidic protein (GFAP), an astrocyte structural protein, has been detected in serum within a few hours after head injury.10 In addition, GFAP has been shown to reliably distinguish between trauma patients with mTBI and those without head injury.10 Blood GFAP levels were elevated in patients with traumatic intracranial abnormalities on CT compared to patients without lesions.10 GFAP could also be used to predict those patients who required neurosurgical intervention.10
Classification performance for detecting intracranial lesions on CT at a GFAP-BDP cutoff level of 0.035 ng/mL yielded a sensitivity of 97% (95%CI 82–100), a specificity of 18% (95%CI 11–28), and a negative predictive value of 94% (95%CI 68–100).10



S100 calcium-binding protein B (S100β) is a calcium-binding protein highly abundant in the astroglia cells of the brain. Several studies have found significant correlation between elevated blood levels of S100β and abnormalities on CT imaging of the brain.14-16 Elevated serum levels of S100β have been associated with an increased incidence of post-concussion syndrome and transient impairment of cognition following trauma.14,17 When the duration of elevated S100β levels was assessed, values decreased at more than 6 hours after injury.18
A drawback of S100β is that it has poor sensitivity (61%) and specificity (77%),19 which is likely to limit its clinical utility in the emergency setting.7 Elevated S100β has been found in patients with fractures and other extracranial injuries.20-22 Additionally, various types of physical activities affect S100β concentrations in apparently healthy athletes.23 One other potential factor is that skin color has been found to affect S100β concentrations, which study authors believe may be related to increased expression of this protein by healthy melanocytes.23,24



Cerebrospinal fluid (CSF) levels of cleaved Tau (c-Tau) protein molecules are significantly elevated following TBI; however, c-Tau levels in peripheral blood do not correlate with the presence of traumatic lesions on CT. This is likely to limit its usefulness in the diagnosis of mTBI in the ED.11,12



Spectrin breakdown product (SBDP) levels are raised in peripheral blood in moderate and severe TBI, yet this increase is not seen in patients with mTBI melanocytes.25



mTBI patients exhibited a significant increase in the serum levels of hyperphosphorylated neurofilaments (p-NF) on days 1 (P<0.001) and 3 (P<0.001),25 however, a 6-hour lag between the onset of injury and the rise in blood levels of p-NF may limit the usefulness of this biomarker as an aid to diagnosis in the acute setting.7
A growing body of research has shown that some blood-based neuro-biomarkers have more clinical utility than others. Three biomarkers that have been extensively studied and associated with TBI are, in astroglia, S100β and GFAP, and in neurons, UCH-L1.
S100β:
GFAP & UCHL-1
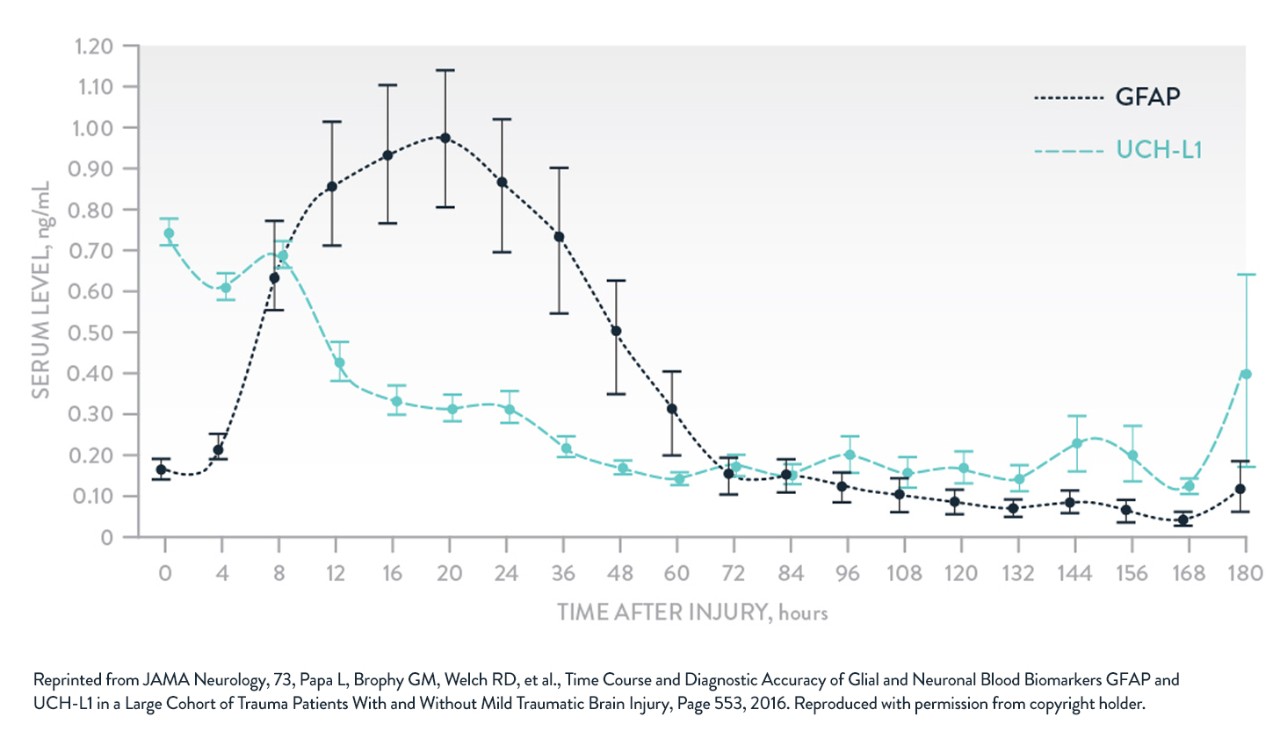
Adapted from Papa et al. JAMA Neurol. 2016.
References:
1. Papa L. Exploring the role of biomarkers for the diagnosis and management of traumatic brain injury patients. INTECH Open Access Publisher. 2012. Available at: https://www.intechopen.com/books/proteomics-human-diseases-and-protein-functions/exploring-the-role-of-biomarkers-for-the-diagnosis-and-management-of-traumatic-brain-injury-patients. [Accessed Sept 19, 2018].
2. Yue JK, Yuh EL, Korley FK, et al. Association between plasma GFAP concentrations and MRI abnormalities in patients with CT-negative traumatic brain injury in the TRACK-TBI cohort: a prospective multicentre study. Lancet Neurol. 2019;18(10):953-961. doi: 10.1016/S1474-4422(19)30282-0.
3. Bazarian JJ, Biberthaler P, Welch RD, et al. Serum GFAP and UCH-L1 for prediction of absence of intracranial injuries on head CT (ALERT-TBI): a multicenter observational study. Lancet Neurol. 2018;17(9):782-789.
4. Michelson EA, Huff JS, Loparo M, et al. Emergency department time course for mild traumatic brain injury workup. West J Emerg Med. 2018;19(4):635-640.
5. Korley FK, Kelen GD, Jones CM, et al. Emergency department evaluation of traumatic brain injury in the united states, 2009–2010. J Head Trauma Rehabil. 2015;31(6):379-387.
6. Zetterberg H, Blennow K. Fluid biomarkers for mild traumatic brain injury and related conditions. Nat Rev Neurol. 2016;12(10):563-574.
7. Jones A, Jarvis P. Review of the potential use of blood neuro-biomarkers in the diagnosis of mild traumatic brain injury. Clin Exp Emerg Med. 2017;4(3):121-127.
8. Wang KK, Yang Z, Zhu T, et al. An update on diagnostic and prognostic biomarkers for traumatic brain injury. Expert Rev Mol Diagn. 2018;18(2):165-180.
9. Ergun R, Bostanci U, Akdemir G, et al. Prognostic value of serum neuron-specific enolase levels after head injury. Neurol Res. 1998;20(5):418-420.
10. Skogseid IM, Nordby HK, Urdal P,et al. Increased serum creatine kinase BB and neuron specific enolase following head injury indicates brain damage. Acta Neurochir. 1992;115:106-111.
11. Ross SA, Cunningham RT, Johnston CF, et al. Neuron-specific enolase as an aid to outcome prediction in head injury. Br J Neurosurg. 1996;10(5):471-476.
12. Verfaillie CJ, Delangh JR. Hemolysis correction factor in the measurement of serum neuron-specific enolase. Clin Chem Lab Med. 2010;48(6):891-892.
13. Kulbe JR, Geddes JW. Current status of fluid biomarkers in mild traumatic brain injury. Exp Neurol. 2016;275:334-335.
14. Ingebrigtsen T, Romner B, Marup-Jensen S, et al. The clinical value of serum S-100 protein measurements in minor head injury: a Scandinavian multicentre study. Brain Injury. 2009;14(12):1047-1055. doi: 10.1080/02699050050203540.
15. Biberthaler P, Linsenmeier U, Pfeifer KJ, et al. Serum S100B concentration provides additional information for the indication of computed tomography in patients after minor head injury. Shock. 2006;25(5):446-453.
16. Muller K, Townend W, Biasca N, et al. S100B serum level predicts computed tomography findings after minor head injury. J Trauma. 2007;62:1452-1456.
17. Waterloo K, Ingebrigtsen T, Romner B. Neuropsychological function in patients with increased serum levels of protein S-100 after minor head injury. Acta Neurochir. 1997;139:26-32.
18. Chabok SY, Moghadam AD, Saneei Z, et al. Neuron-specific enolase and S100BB as outcome predictors in severe diffuse axonal injury. J Trauma Acute Care Surg. 2012;72(6):1654-1657.
19. Nygren de boussard C, Fredman P, Lundin A, et al. S100 in mild traumatic brain injury. Brain Inj. 2004;18(7):671-683.
20. Anderson RE, Hansson LO, Nilsson O, et al. High serum S100B levels for trauma patients without head injuries. Neurosurgery. 2001;48(6):1255-1260.
21. Unden J, Bellner J, Eneroth M, et al. Raised serum S100B levels after acute bone fractures without cerebral injury. J Trauma. 2005;58:59-61.
22. Papa L, Silvestri S, Brophy GM, et al. GFAP Out-Performs S100β in Detecting Traumatic Intracranial Lesions on Computed Tomography in Trauma Patients with Mild Traumatic Brain Injury and Those with Extracranial Lesions. J Neurotrauma. 2014;31(22):1815-1822.
23. Schulte S, Podlog LW, Hamson-Utley JJ, et al. A systematic review of the biomarker S100B: implications for sport-related concussion management. J Athl Train. 2014;49(6):830-850.
24. Abdesselam OB, Vally J, Adem C, et al. Reference values for serum S-100B protein depend on the race of individuals [letter to the editor]. Clin Chem. 2003;49(5):836-837.
25. Gatson JW, Barillas J, Hynan LS, et al. Detection of neurofilament-H in serum as a diagnostic tool to predict injury severity in patients who have suffered mild traumatic brain injury. J Neurosurg. 2014;121(5):1232-1238.
26. Andres R, Mayordomo JI, Zaballos P, et al. Prognostic value of serum S-100B in malignant melanoma. Tumori. 2004;90:607-610.
27. Su YRS, Schuster JM, Smith DH, et al. Cost-effectiveness of biomarker screening for traumatic brain injury. J Neurotrauma. 2019;36:2083-2091.
28. Papa L, Brophy GM, Welch RD, et al. Time course and diagnostic accuracy of glial and neuronal blood biomarkers GFAP and UCH-L1 in a large cohort of trauma patients with and without mild traumatic brain injury. JAMA Neurol. 2016;73(5):551-560.
29. Diaz-Arrastia R, Wang KKW, Papa L, et al. Acute biomarkers of traumatic brain injury: relationship between plasma levels of ubiquitin C-terminal hydrolase-L1 and glial fibrillary acidic protein. J Neurotrauma. 2014;31(1):19-25.
30. Takala RSK, Posti JP, Runtti H, et al. Glial fibrillary acidic protein and ubiquitin c-terminal hydrolase-L1 as outcome predictors in traumatic brain injury. World Neurosurg. 2016;87:8-20.
Links which take you out of Abbott worldwide websites are not under the control of Abbott, and Abbott is not responsible for the contents of any such site or any further links from such site. Abbott is providing these links to you only as a convenience, and the inclusion of any link does not imply endorsement of the linked site by Abbott. The website that you have requested also may not be optimised for your screen size.
Click yes if you are a healthcare professional.